Microfluidics: Putting Biological Research on a Chip
It’s been two big years for Microfluidics. Now MEMS-based Organs-on-a-chip and other biological simulations are showing huge promise in medical applications.
Humans like to use metaphors. At minimum, it’s more fun. And in the best of times, it can help us understand things that are quite complicated on their own.[i]
The challenge is that these metaphors are often wrong.
While we may not be able to understand very complex systems today, there is a lot we can do to build up to that. We may not have a model for how the universe formed, but we can use CERN’s large hadron supercollider to build up knowledge of how it might have happened. Instead of a linguistic metaphor, we built a physical one.
We’re also doing the same thing with biology, designing, and building physical models of organisms that were neither designed nor built. If we can understand organisms or parts of organisms as if they were designed to fulfill a specific purpose, it may point us in the right direction, advancing our understanding of how they operate in the real world.
[i] Living beings are very unlike systems designed by humans. The human brain is not at all like a computer. The immune system is only slightly less complicated, and it’s still “Where Intuition Goes to Die.” According to evolutionary theory, biological systems and components are not shaped by specific purpose they’re meant to fulfill. They’re the result of a complex interplay of chemistry and physics, sometimes brilliantly simple, and other times seemingly contradictory or even self-defeating.
Microfluidics – New Technologies and Applications for MEMS
Increasingly, tools that model biological systems rely on microelectromechanical systems (MEMS). These systems are made up of components ranging from 1 to 100 micrometers, with MEMS devices ranging from 20 micrometers to a millimeter (although some, like micromirror arrays used sometimes in lidar systems can be much larger). They typically include an integrated circuit chip for data processing and then sensors for collecting data (such as gyroscopes, accelerometers, microphones) and then actuators that allow the device to respond to sensor inputs.
MEMS can also be used in microfluidics, which is the design and use of devices that manipulate and measure tiny amounts of fluids, usually in the range of microliters and pico-liters. These microfluidic MEMS chips are made by etching thin grooves or small wells on the surface of a layer, and then enclosing those features by means of a second layer to form microchannels or chambers that can direct these tiny amounts of fluid.
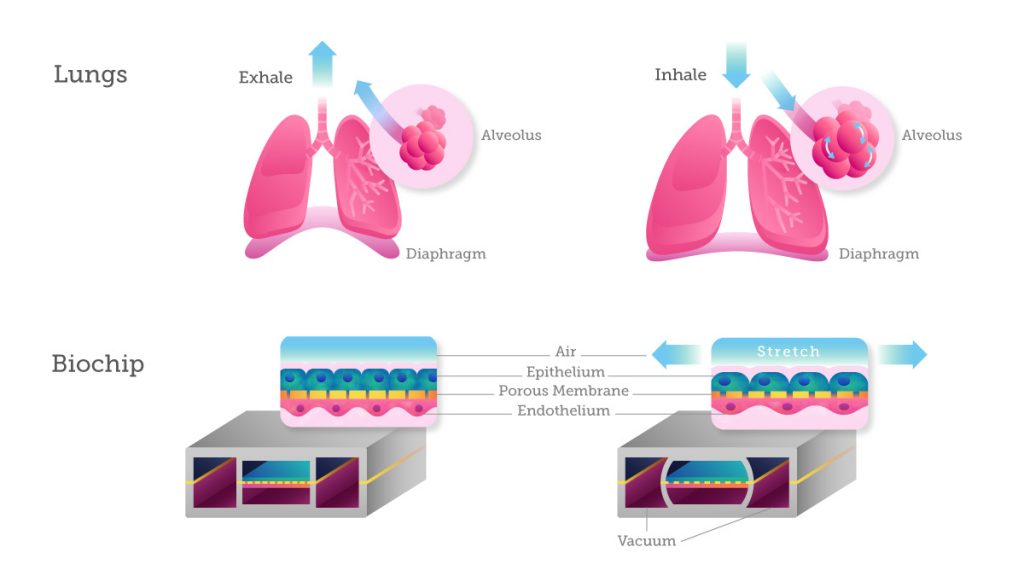
While it’s simple in concept, the materials, size, and required accuracy make MEMS microfluidics very challenging. Assembly, integration, and packaging must be very precise, aligning fluidic, optical, and electronic pathways perfectly to endure harsh environments and pressures. Errors in design or manufacturing mean creating a device that isn’t sufficiently reliable. In 2020, industry analysts such as Yole Development predicted that many exciting microfluidic applications, still needed another two decades or more of development to achieve truly widespread adoption.
With Covid-19, that outlook may have changed. Medical approvals such as emergency use authorization by the FDA and CE marking were simplified. In response, several new players reached the commercialization stage of their products. The pandemic has boosted the global microfluidics market by 45% in two years, from US$9.9 billion in 2019 to US$18.1 billion in 2021, driven by wider adoption of point-of-care solutions and next-generation sequencing (NGS) technologies. This growth has energized the industry, with more people looking at where they can employ microfluidic MEMS devices as cost-effective tools for biomedical research, cell biology, and drug discovery.
Making Microfluidic MEMS imitate biology
One of the most exciting applications may be what’s called “organ-on-a-chip,” where MEMS system is created to replicate functional kidney, brain, and heart tissues. This can help researchers study of molecular mechanisms of action for various treatments and more quickly prioritize lead candidates, perform toxicity testing, and identify biomarkers.
Today organ-on-a-chip devices can model both normal physiology and disease states of human organs (e.g., lung, intestine, kidney, bone marrow), from fluid flows, physical structure, and even cellular composition, which affect cell development and tissue function.
For example, researchers at Harvard’s Wyss Institute were able to model parts of the human immune system. In the past, “Animals have been the gold-standard research models for developing and testing new vaccines, but their immune systems differ significantly from our own and do not accurately predict how humans will respond to them.” Says Girija Goyal, Ph.D., a Senior Staff Scientist at the Wyss Institute. Their lymphoid follicle chip was a way to model the complex sequence of human immune responses to infection and vaccination. They hope that it could improve the speed and success of vaccine development in the future.
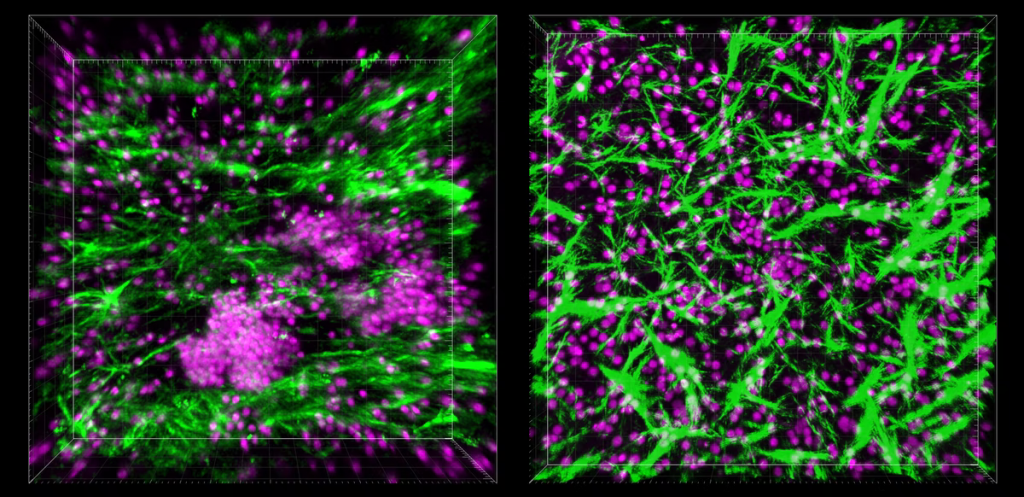
The flexibility of the devices means that not only can they be more accurate than animal analogs, but also adapted to better capture information in real-time. Real-time imaging of living multicellular organisms is challenging – the subject is very small and in motion. Some researchers have found innovative ways to slow down or otherwise alter processes to make them more visible without interfering with the results.
Others are looking at ways to integrate vision systems into the MEMS chips. Today, researchers have reported the integration of microfluidics with CMOS sensors for cell analysis, and have even been successful at detecting various diseases, such as malaria, influenza, hepatitis B virus, COVID-19, and bacteria-based diseases.
What’s Next?
New levels of investment in microfluidics will certainly see growth in point-of-care diagnostic solutions, with hundreds of million units per year already. There remains enormous growth opportunities in places without robust medical infrastructure, such as developing countries and remote locations.
Non-invasive ways to diagnose and monitor cancer are still at the research phase, but represent a potent possibility for more responsive and accurate treatment, where screening can be done quickly, cheaply, and globally.
Organs-on-chips are just getting started. Their use in drug discovery and development will continue to move us toward more accurate and effective medical care. It will also be more humane, eliminating the need for animal or human test subjects.
CMOS is also expected to play a growing role in microfluidic platforms, leveraging existing visual medical data but further expanding what can be achieved – replacing expensive instruments and diagnostic techniques with integrated vision systems that are cheaper and smaller than we ever thought possible.
[1] Living beings are very unlike systems designed by humans. The human brain is not at all like a computer. The immune system is only slightly less complicated, and it’s still “Where Intuition Goes to Die.” According to evolutionary theory, biological systems and components are not shaped by specific purpose they’re meant to fulfill. They’re the result of a complex interplay of chemistry and physics, sometimes brilliantly simple, and other times seemingly contradictory or even self-defeating.


 MEMS and Micromachines of Guiding Light
MEMS and Micromachines of Guiding Light  You Spin Me Right Round: Measuring Movement with MEMS
You Spin Me Right Round: Measuring Movement with MEMS 